Market Overview:
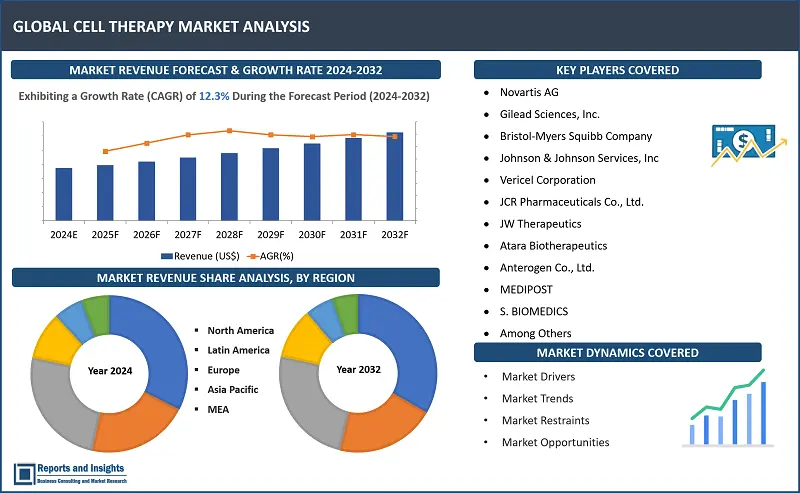
"The global cell therapy market size reached US$ 4.7 billion in 2023. Looking forward, Reports and Insights expects the market to reach US$ 13.4 billion in 2032, exhibiting a growth rate (CAGR) of 12.3% during 2024-2032."
|
Report Attributes |
Details |
|
Base Year |
2023 |
|
Forecast Years |
2024-2032 |
|
Historical Years |
2021-2023 |
|
Market Growth Rate (2024-2032) |
12.3% |
Cell therapy (CT) is a groundbreaking field within regenerative medicine, and procedures involve the transplantation of living human cells to repair or replace damaged tissue and or cells. Different types of cells can be used as part of a therapy or treatment for a variety of diseases and conditions, as well as in sports medicine and severe burn care, and also can be used to prevent potential diseases. CT encompasses various technologies, solutions, and services aimed at harnessing the therapeutic potential of cells to repair, replace, or rejuvenate damaged tissues. Advancements in cell therapy have led to promising breakthroughs in treating conditions such as cancer, autoimmune disorders, and degenerative diseases. The approach often involves manipulating cells, either from the patient's own body (autologous) or from donors (allogeneic), to enhance their therapeutic properties.
Some benefits and advantages of cell therapy include reduced side effects compared to traditional treatments and medication, improved precision in targeting specific cells or tissues, and the potential for long-lasting therapeutic effects. Stem cells, in particular, play a crucial role in many cell therapy applications due to their ability to differentiate into various cell types.
Key trends in the global cell therapy market include increasing investments in Research and Development (R&D), growing partnerships and collaborations among biotech companies, and an inclining focus on personalized medicine. Notable developments in the market include the expansion of regulatory approvals for cell therapy products, such as CAR-T cell therapies for certain types of cancers, indicating a maturing and evolving landscape in the global cell therapy market. As technology continues to advance, cell therapy holds immense promise in transforming the treatment landscape for a wide range of medical conditions.
Cell Therapy Market Trends and Drivers:
Key factors driving growth of the CT market include increasing prevalence of chronic diseases, such as cancer and autoimmune disorders, which has been resulting in increased focus on the exploration of innovative treatment options like cell therapy. Also, advancements in biotechnology and genetic engineering have significantly improved the understanding and manipulation of cellular functions, thereby opening up avenues and possibilities for development of more effective and targeted cell-based treatments.
In addition, the trend towards personalized medicine is a major factor driving the adoption of cell therapy, as the ability to tailor treatments to individual patients, utilizing their own cells, enhances therapeutic outcomes and minimizes adverse effects. Another driving factor is rising investment in research and development by pharmaceutical companies and biotech firms in the discovery of new cell therapy applications to accelerate the development of novel therapies.
Moreover, regulatory approvals for cell therapy products, such as approval for CAR-T cell therapies, is creating a regulatory framework that instills confidence in both practitioners and patients. This supportive regulatory environment is supporting the commercialization of cell therapies, and as more therapies progress from clinical trials to market availability, growth of the global cell therapy market is expected to continue to gain substantial traction.
Cell Therapy Market Restraining Factors:
Factors such as manufacturing complexities and the associated high costs limit scalability, and these coupled with logistical challenges in handling and transporting living cells contribute to operational hurdles. Also, concerns regarding the potential for adverse reactions or long-term side effects in patients, especially with allogeneic cell therapies, create apprehension among healthcare providers and regulators. In addition, limited reimbursement policies and uncertain reimbursement models pose financial challenges for both patients and healthcare systems. The regulatory landscape remains dynamic, with evolving standards that can slow down approvals and market entry, and these are having a negative impact on overall growth of the market.
Negative trends in the market include skepticism among investors due to prolonged timelines for returns on investments and market volatility. Challenges such as need for standardized manufacturing processes and addressing ethical concerns also contribute to a cautious approach. Navigating these obstacles will be essential to unlocking the full potential of cell therapy and achieving sustained market growth.
Cell Therapy Market Opportunities:
Leading companies in the global cell therapy market have several lucrative opportunities including expansion of indications for existing cell therapies. As research advances, the application of cell therapies could broaden beyond oncology to include neurodegenerative disorders, cardiovascular diseases, and other. Also, strategic collaborations and partnerships offer opportunities for companies to leverage complementary expertise, enhance research capabilities, and accelerate product development.
Moreover, the rise of the personalized medicine trend is opening up avenues for companies to develop tailored cell therapies, capitalizing on patient-specific treatments that can improve efficacy and reduce adverse effects. In addition, the increasing focus on regenerative medicine and the use of stem cells in cell therapy provide opportunities for innovative therapeutic approaches. Furthermore, investments in process optimization and automation technologies can streamline manufacturing, reducing costs and enhancing scalability, ultimately resulting in wider market access.
Cell Therapy Market Segmentation:
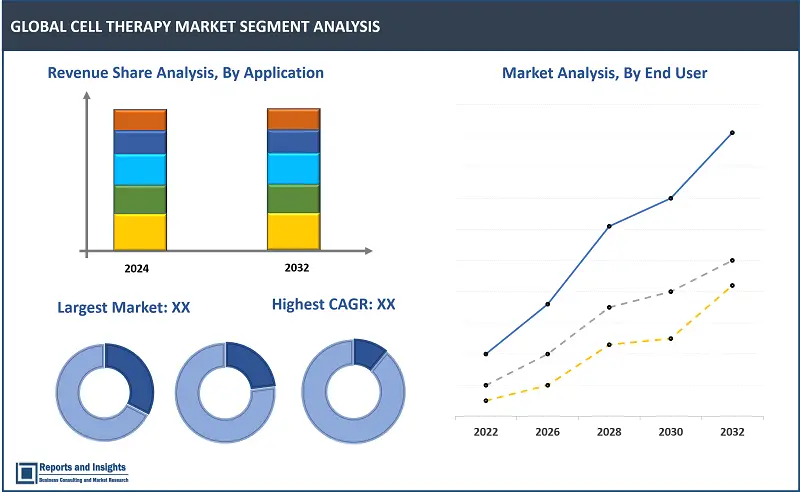
By Type of Cell Therapy
- Autologous Cell Therapy
- Allogeneic Cell Therapy
The autologous cell therapy segment is expected to account for largest revenue share among the cell therapy type segments over the forecast period. This can be attributed to high preference for this type owing to the inherent advantages of using a patient's own cells, thereby minimizing risk of rejection and adverse reactions. Also, the personalized nature of autologous cell therapy aligns with the growing trend towards individualized medicine.
By Application
- Oncology
- Cardiovascular Disorders
- Musculoskeletal Disorders
- Other Therapeutic Areas
The oncology segment is projected to dominate other application segments in terms of revenue share in the global cell therapy market. This is primarily due to the extensive R&D efforts focused on developing cell therapies for various cancers, including hematological malignancies and solid tumors. The success of CAR-T cell therapies in treating certain types of leukemia and lymphoma has driven significant interest and investment in oncology-related cell therapies. Also, increasing prevalence of cancer worldwide is further driving wide adoption in oncology applications.
By Cell Source
- Stem Cell-based Therapies
- Non-Stem Cell-based Therapies
Among the cell source segments, the stem cell-based therapies segment is expected to account for largest revenue share, driven the versatile nature of stem cells, offering the potential to differentiate into various cell types for therapeutic applications. Stem cells, with their regenerative properties, hold potential for addressing a wide range of medical conditions, and this coupled with ongoing R&D in stem cell technologies is contributing to increased adoption.
By End User
- Hospitals and Clinics
- Research Institutes
- Biotechnology and Pharmaceutical Companies
The hospital and clinics segment is expected to account for largest revenue share among the end-user segments over the forecast period. This can be attributed to increasing integration of cell therapies into mainstream medical practices and treatment protocols within healthcare facilities. Hospitals and clinics serve as primary points of care, facilitating the administration and management of cell therapies. The growing awareness, expanding infrastructure, and accessibility of these therapies within clinical settings is expected to continue to contribute to growth of the hospital and clinics segment over the forecast period.
By Region
North America
- United States
- Canada
Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Russia
- Poland
- Benelux
- Nordic
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Latin America
- Brazil
- Mexico
- Argentina
Middle East & Africa
- Saudi Arabia
- South Africa
- United Arab Emirates
- Israel
- Rest of MEA
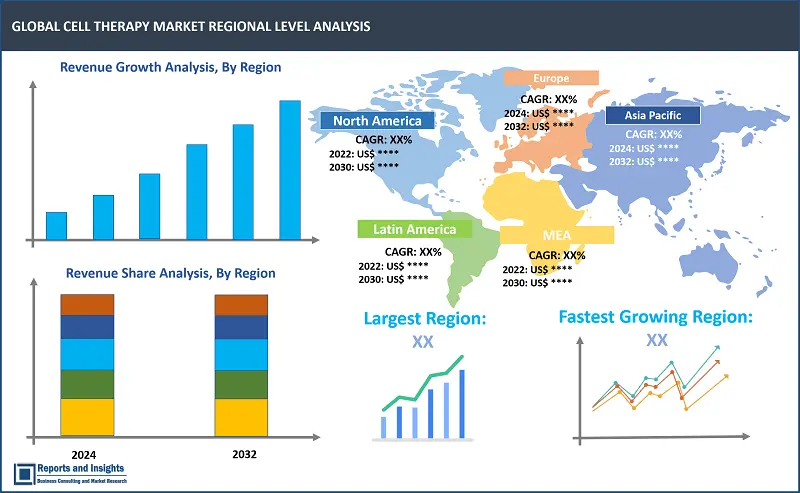
The global Cell Therapy Market is divided into five key regions: North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. North America market growth has been significantly robust, with majority revenue share contribution from the market in the United States (US), attributed to a robust healthcare infrastructure, substantial research and development initiatives, and supportive regulatory frameworks. In Europe, countries such as Germany and the United Kingdom account for significant revenue contribution owing to presence of advanced healthcare systems and key focus on innovation. Asia Pacific has also been registering steady revenue growth, with countries such as Japan and China accounting substantially due to incline in initiatives and research that are cell therapy-based, as well as increasing investments in biotechnology and healthcare research.
Some common factors driving overall growth in these regional markets include increasing prevalence of chronic diseases, rising investment in R&D, and growing emphasis on personalized medicine.
Leading Companies in Cell Therapy & Competitive Landscape:
The competitive landscape in the global cell therapy market is dynamic and characterized by the presence of key players employing various strategies to maintain their positions and expand their consumer bases. Leading companies are investing substantially in research and development to advance their cell therapy technologies and broaden the range of treatable conditions. Strategic collaborations and partnerships are also prevalent, facilitating the sharing of expertise, resources, and the acceleration of product development. Also, mergers and acquisitions play a crucial role in strengthening market presence and diversifying product portfolios. Companies are actively engaging in commercialization efforts, seeking regulatory approvals and expanding their geographical reach to tap into emerging markets.
These companies include:
- Novartis AG
- Gilead Sciences, Inc.
- Bristol-Myers Squibb Company
- Johnson & Johnson Services, Inc
- Vericel Corporation
- JCR Pharmaceuticals Co., Ltd.
- JW Therapeutics
- Atara Biotherapeutics
- Anterogen Co., Ltd.
- MEDIPOST
- S. BIOMEDICS
- Aurion Biotech
- Holostem Terapie Avanzate S.r.l
- Nkarta, Inc.
- Adaptimmune Therapeutics plc
Recent Development:
- February 05, 2024: Novartis announced entering in agreement for a voluntary public takeover of MorphoSys AG, which is a global biopharmaceutical company based in Germany, specializing in innovative oncology medicines. The acquisition, contingent upon customary closing conditions and regulatory approvals, is expected to boost Novartis' oncology pipeline, add key therapeutic focus, and expand its global presence in hematology. Upon completion of acquisition, Novartis will gain control of pelabresib (CPI-0610), which is a potentially transformative treatment in combination with ruxolitinib for myelofibrosis (MF) patients, and tulmimetostat (CPI-0209), an early-stage dual inhibitor of enhancer of zeste homolog 1 and 2 (EZH1 and EZH2) proteins under investigation for solid tumors or lymphomas. Pelabresib, in combination with ruxolitinib, recently demonstrated positive outcomes in Phase 3 MANIFEST-2 trials, meeting primary and secondary endpoints, showcasing its potential as a myelofibrosis treatment. The planned acquisition underscores Novartis' commitment to fortifying its position in oncology through strategic portfolio enhancements.
- January 30, 2024: Kite, which is a subsidiary of Gilead, announced that the U.S. Food and Drug Administration (FDA) has sanctioned a modification in the manufacturing process, resulting in a reduced manufacturing time for Yescarta (axicabtagene ciloleucel). With this regulatory approval, Kite anticipates a decrease in its median turnaround time in the U.S. from 16 days to 14 days. The median TAT encompasses the period from leukapheresis, where a patient's T cells are collected, to product release. The manufacturing adjustment is a pivotal step in this process, crucial for preparing a patient's cells for a personalized, one-time cell therapy infusion. Yescarta stands out as the initial and exclusive treatment demonstrating superior overall survival compared to the standard of care in second-line treatment with curative intent. The FDA's approval enables Kite to further streamline Yescarta's delivery time, aiming to enhance patient chances of survival.
- May 2023: Johnson & Johnson announced that its antibody-based therapy for a challenging-to-treat form of blood cancer had received approval from the US Food and Drug Administration. Marketed as Talvey, the therapy, talquetamab-tgvs, falls under the category of bispecific antibodies. This class of treatments is engineered to facilitate the interaction between a cancer cell and an immune cell, representing a significant advancement in the treatment of this particular type of blood cancer.
Cell Therapy Market Research Scope
|
Report Metric |
Report Details |
|
Market size available for the years |
2021-2023 |
|
Base Year |
2023 |
|
Forecast Period |
2024-2032 |
|
Compound Annual Growth Rate (CAGR) |
12.3% |
|
Segment covered |
Type of Cell Therapy, Application, Cell Source, End-Users |
|
Regions Covered |
North America: The U.S. & Canada Latin America: Brazil, Mexico, Argentina, & Rest of Latin America Asia Pacific: China, India, Japan, Australia & New Zealand, ASEAN, & Rest of Asia Pacific Europe: Germany, The U.K., France, Spain, Italy, Russia, Poland, BENELUX, NORDIC, & Rest of Europe The Middle East & Africa: Saudi Arabia, United Arab Emirates, South Africa, Egypt, Israel, and Rest of MEA |
|
Fastest Growing Country in Europe |
UK |
|
Largest Market |
North America |
|
Key Players |
Novartis AG, Gilead Sciences, Inc., Bristol-Myers Squibb Company, Johnson & Johnson Services, Inc., Vericel Corporation, JCR Pharmaceuticals Co., Ltd., JW Therapeutics, Atara Biotherapeutics, Anterogen Co., Ltd., MEDIPOST, S. BIOMEDICS, Aurion Biotech, Holostem Terapie Avanzate S.r.l, Nkarta, Inc., Adaptimmune Therapeutics plc. |
Frequently Asked Question
What is the size of the global cell therapy market in 2022?
The global cell therapy market size reached US$ 4.7 Billion in 2023.
At what CAGR will the global cell therapy market expand?
The global market is expected to register a 12.3% CAGR through 2024-2032.
Who is the leader in the global cell therapy market?
Gilead Sciences, Inc. is a leader in the global cell therapy market.
What are some key factors driving revenue growth of the cell therapy market?
Factors contributing to revenue growth of the cell therapy market include increasing prevalence of chronic diseases, advancements in biotechnology, growing investments in research and development, expanding applications in oncology, and a rising trend towards personalized medicine.
What are some major challenges faced by companies in the cell therapy market?
Companies in the cell therapy market encounter challenges such as manufacturing complexities, logistical issues in handling living cells, concerns about adverse reactions, reimbursement challenges, and navigating the evolving regulatory landscape. Also, scepticism among investors and the need for standardized processes pose significant hurdles.
How is the competitive landscape in the cell therapy market?
The competitive landscape in the cell therapy market is dynamic, featuring key players like Novartis, Gilead Sciences, and others. Intense competition drives companies to invest in research, form strategic collaborations, engage in mergers and acquisitions, and focus on commercialization efforts to maintain a competitive edge.
How is the global cell therapy market report segmented?
The global cell therapy market report typically includes segmentation based on the type of cell therapy (autologous, allogeneic), application (oncology, cardiovascular disorders, musculoskeletal disorders, etc.), cell source (stem cell-based, non-stem cell-based), and end-users (hospitals and clinics, research institutes, biotechnology and pharmaceutical companies).
Who are the key players in the global cell therapy market report?
Key players in the global cell therapy market report include Novartis AG, Gilead Sciences, Inc., Bristol-Myers Squibb Company, Johnson & Johnson Services, Inc., Vericel Corporation, JCR Pharmaceuticals Co., Ltd., JW Therapeutics, Atara Biotherapeutics, Anterogen Co., Ltd., MEDIPOST, S. BIOMEDICS, Aurion Biotech, Holostem Terapie Avanzate S.r.l, Nkarta, Inc., Adaptimmune Therapeutics plc.

