Market Overview:
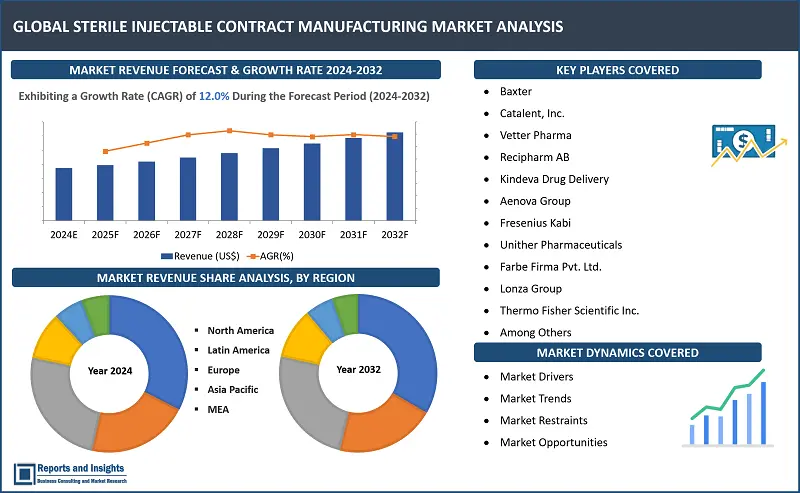
"The global sterile injectable contract manufacturing market size reached US$ 16.2 billion in 2023. Looking forward, Reports, Insights expects the market to reach US$ 40.0 billion by 2032, exhibiting a growth rate (CAGR) of 12.0% during 2024-2032."
|
Report Attributes |
Details |
|
Base Year |
2023 |
|
Forecast Years |
2024-2032 |
|
Historical Years |
2021-2023 |
|
Market Growth Rate (2024-2032) |
12.0% |
Sterile injectable contract manufacturing involves outsourcing the production of sterile injectable pharmaceuticals to specialized third-party manufacturers. These manufacturers have the facilities and expertise needed to produce sterile drugs like vaccines, biologics, and injectable medications, meeting stringent regulatory standards. Contract manufacturing organizations (CMOs) offer services such as formulation development, sterile filling, packaging, and quality control testing. This model enables pharmaceutical companies to concentrate on research, marketing, and distribution, providing flexibility, scalability, and cost-effectiveness in bringing sterile injectable products to market.
The sterile injectable contract manufacturing market is experiencing significant growth due to rising global demand for sterile injectable pharmaceuticals. Pharmaceutical firms are increasingly subcontracting the production of sterile injectable medications to specialized contract manufacturing organizations (CMOs) to lower costs, enhance efficiency, and gain access to specialized expertise and facilities. Factors such as the increasing prevalence of chronic diseases, the expansion of the biopharmaceutical industry, and the demand for innovative drug delivery systems are driving market expansion. Furthermore, stringent regulatory requirements and the need to comply with Good Manufacturing Practices (GMP) are boosting the demand for sterile injectable contract manufacturing services. The market is characterized by technological advancements, strategic collaborations, and a focus on developing novel formulations and delivery systems to meet the evolving needs of the healthcare sector.
Sterile Injectable Contract Manufacturing Market Trends and Drivers:
The sterile injectable contract manufacturing market is shaped by several trends and drivers. A notable trend is the increasing reliance of pharmaceutical companies on specialized contract manufacturing organizations (CMOs) for sterile injectable production, driven by cost reduction and operational efficiency goals. The market is further propelled by the rising demand for injectable medications, especially for chronic diseases and cancer treatments. Technological advancements, particularly in aseptic filling and lyophilization processes, are also enhancing manufacturing efficiency and product quality. Regulatory compliance, notably with guidelines like Good Manufacturing Practices (GMP), is a critical focus, prompting companies to adopt advanced manufacturing practices for ensuring product safety and quality. These dynamics collectively point to a favorable outlook for the sterile injectable contract manufacturing market, indicating sustained growth in the foreseeable future.
The sterile injectable contract manufacturing market growth is influenced by several factors which include growing demand for sterile injectable pharmaceuticals, fueled by the increasing prevalence of chronic diseases and the consequent need for more effective treatment options. The market is further propelled by the complexity of manufacturing processes, which necessitate specialized facilities and expertise, prompting pharmaceutical companies to outsource production to contract manufacturing organizations (CMOs). Technological advancements in aseptic processing and lyophilization techniques are also key growth drivers, enabling CMOs to produce high-quality sterile injectable products. Additionally, stringent regulatory requirements, including compliance with Good Manufacturing Practices (GMP), are pushing pharmaceutical companies to collaborate with CMOs that can meet these standards. These factors, coupled with the rising adoption of contract manufacturing services by pharmaceutical companies, are expected to fuel the growth of the sterile injectable contract manufacturing market in the coming future.
Sterile Injectable Contract Manufacturing Restraining Factors:
The growth of the sterile injectable contract manufacturing market is constrained by several factors. A major factor is the substantial capital investment required to establish and maintain sterile manufacturing facilities, including cleanrooms and specialized equipment. This can discourage new entrants and limit the expansion of existing players. Additionally, the complex regulatory environment and the need to adhere to stringent quality standards like GMP present challenges for contract manufacturers, leading to higher operational costs and potential delays in product development and approval. Furthermore, the scarcity of skilled personnel with expertise in sterile injectable manufacturing poses a constraint, affecting the ability of CMOs to scale up operations and meet increasing demand. These factors, along with market competition and pricing pressures from pharmaceutical companies, contribute to the difficulties faced by players in the sterile injectable contract manufacturing market.
Sterile Injectable Contract Manufacturing Market Opportunities:
The sterile injectable contract manufacturing market offers several growth opportunities. One significant prospect is the rising demand for specialized injectable therapies, particularly in oncology, autoimmune diseases, and biologics. Contract manufacturing organizations (CMOs) can leverage this trend by providing flexible and cost-effective manufacturing solutions to pharmaceutical firms seeking to introduce new injectable medications. Moreover, technological advancements like robotics and automation in manufacturing processes present opportunities for CMOs to enhance efficiency and reduce costs. Additionally, the increasing shift toward personalized medicine and the development of complex biologic drugs are expected to boost demand for specialized manufacturing capabilities, opening up new avenues for CMOs to diversify their services and expand their market presence.
Sterile Injectable Contract Manufacturing Market Segmentation:
By Molecule Type
- Small Molecule
- Large Molecule
In the sterile injectable contract manufacturing market, the large molecule sub-segment currently holds the dominant position. Large molecules, such as biologics and complex protein-based drugs, require specialized manufacturing processes and facilities, leading to their outsourcing to contract manufacturing organizations (CMOs). The increasing demand for biologics, due to their efficacy in treating various diseases, is a key driver for this sub-segment's growth. Moreover, advancements in biotechnology and the development of innovative biologic drugs are further boosting the demand for large molecule contract manufacturing services. Despite this dominance, the small molecule sub-segment remains significant, especially for traditional injectable medications, and is expected to grow alongside the large molecule sub-segment in the future.
By Therapeutic Application
- Cancer
- Diabetes
- Cardiovascular Diseases
- Central Nervous System Diseases
- Infectious Disorders
- Musculoskeletal
- Anti-viral
- Others
The cancer therapeutic application sub-segment currently holds the dominant position in the sterile injectable contract manufacturing market. This is driven by the increasing global prevalence of cancer and the rising demand for advanced cancer treatments, including injectable therapies. Contract manufacturing organizations (CMOs) play a vital role in meeting this demand by offering specialized manufacturing services for cancer drugs like chemotherapy agents and immunotherapies. Furthermore, ongoing advancements in cancer research and the development of targeted therapies are contributing to the growth of contract manufacturing services in this sub-segment. While cancer is the dominant player, other therapeutic areas such as diabetes, cardiovascular diseases, and infectious disorders also make significant contributions to the market and are expected to see growth in the future.
By Route of Administration
- Subcutaneous (SC)
- Intravenous (IV)
- Intramuscular (IM)
- Others
The intravenous (IV) route of administration sub-segment currently holds the dominant position in the sterile injectable contract manufacturing market. IV injections are widely utilized for administering medications directly into the bloodstream, enabling rapid onset of action and precise dosing. This route is commonly employed for various injectable pharmaceuticals, including antibiotics, chemotherapy drugs, and biologics. Contract manufacturing organizations (CMOs) play a pivotal role in providing sterile IV drug manufacturing services, meeting the substantial demand from pharmaceutical firms. While the IV route is dominant, other administration routes such as subcutaneous (SC), intramuscular (IM), and others also play significant roles in the market and are anticipated to grow in importance with the development of new therapies and delivery methods.
By End-use
- Pharmaceutical Companies
- Biopharmaceutical Companies
- Others
In the sterile injectable contract manufacturing market, the pharmaceutical companies end-use segment currently holds the dominant position. These companies are increasingly opting to outsource the production of sterile injectable drugs to specialized contract manufacturing organizations (CMOs) to cut costs and focus on their core competencies such as research and development. Pharmaceutical firms require CMOs to meet stringent regulatory standards and manufacture high-quality sterile injectable products, driving the demand for contract manufacturing services. The biopharmaceutical companies sub-segment also plays a significant role in the market, particularly in the production of biologics and complex injectable therapies. While other end-use segments contribute to the market, they are not as dominant as pharmaceutical and biopharmaceutical companies.
By Region
North America
- United States
- Canada
Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Russia
- Poland
- Benelux
- Nordic
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
Latin America
- Brazil
- Mexico
- Argentina
Middle East & Africa
- Saudi Arabia
- South Africa
- United Arab Emirates
- Israel
- Rest of MEA
North America currently leads the sterile injectable contract manufacturing market due to several key factors. The well-established pharmaceutical and biotechnology sector in the region drives the demand for sterile injectable drugs, especially with the increasing prevalence of chronic diseases and aging populations. North America's stringent regulatory environment ensures high standards of quality and safety, making it a preferred location for contract manufacturing organizations (CMOs). Additionally, the region's advanced technology in aseptic processing and lyophilization techniques, along with the presence of major market players, further solidify its dominance in the market. Factors such as its strategic location, skilled workforce, and robust infrastructure all contribute to North America's strong position in sterile injectable contract manufacturing.
Leading Sterile Injectable Contract Manufacturing Manufacturers & Competitive Landscape:
The sterile injectable contract manufacturing market is highly competitive, with several key players vying for market share and actively engaging in strategic initiatives. These companies focus on product innovation, technological advancements, and expanding their product portfolios to gain a competitive edge. These companies are continuously investing in research and development activities to enhance their product offerings and cater to the evolving needs of customers in terms of efficiency, performance, and sustainability.
These companies include:
- Baxter
- Catalent, Inc.
- Vetter Pharma
- Recipharm AB
- Kindeva Drug Delivery
- Aenova Group
- Fresenius Kabi
- Unither Pharmaceuticals
- Farbe Firma Pvt. Ltd.
- Lonza Group
- Thermo Fisher Scientific Inc.
- Pfizer CentreOne
- AbbVie Contract Manufacturing
- Jubilant HollisterStier
- CordenPharma International
- Fareva
- Among Others
Recent News and Development
- February 2024: Simtra BioPharma Solutions has announced a significant investment exceeding $250 million to expand its sterile fill/finish manufacturing campus in Bloomington, Indiana.
- April 2023: Bridgewest Group, known for its innovation in life sciences, software, and deep tech sectors, has launched a new Contract Development and Manufacturing Organization (CDMO) specializing in sterile injectable drug products. This initiative follows the recent acquisition of Pfizer's sterile injectable manufacturing plant in Perth.
Sterile Injectable Contract Manufacturing Market Research Scope
|
Report Metric |
Report Details |
|
Market size available for the years |
2021-2023 |
|
Base Year |
2023 |
|
Forecast Period |
2024-2032 |
|
Compound Annual Growth Rate (CAGR) |
12.0% |
|
Segment covered |
Molecule type, therapeutic application, route of administration, end-use and regions. |
|
Regions Covered |
North America: The U.S. Canada Latin America: Brazil, Mexico, Argentina, & Rest of Latin America Asia Pacific: China, India, Japan, Australia & New Zealand, ASEAN, & Rest of Asia Pacific Europe: Germany, The U.K., France, Spain, Italy, Russia, Poland, BENELUX, NORDIC, & Rest of Europe The Middle East & Africa: Saudi Arabia, United Arab Emirates, South Africa, Egypt, Israel, and Rest of MEA |
|
Fastest Growing Country in Europe |
Germany |
|
Largest Market |
North America |
|
Key Players |
Baxter, Catalent, Inc., Vetter Pharma, Recipharm AB, Aenova Group, Fresenius Kabi, Unither Pharmaceuticals, Famar, Cipla Inc. and NextPharma Technologies. |
Frequently Asked Question
At what CAGR will the sterile injectable contract manufacturing market expand?
The market is anticipated to rise at 12.0% through 2032.
Who are the major end-use of sterile injectable contract manufacturing market?
The major end-users of sterile injectable contract manufacturing market are pharmaceutical companies, biopharmaceutical companies, and others.
Which region accounted for the largest market share in 2023?
North America region accounted for the largest market share in 2023
What are some key factors driving revenue growth of the sterile injectable contract manufacturing market?
Some key factors driving sterile injectable contract manufacturing market revenue growth include increasing demand for sterile injectable drugs, advancements in biotechnology, outsourcing trends, technological advancements, and market expansion.
What are some major challenges faced by companies in the sterile injectable contract manufacturing market?
Companies face challenges such as stringent regulatory requirements, high capital investment, quality control and assurance, supply chain disruptions, and the changing healthcare landscape.
How is the competitive landscape in the sterile injectable contract manufacturing market?
The market is competitive, with key players focusing on technological advancements, product innovation, and strategic partnerships. Factors such as product quality, reliability, after-sales services, and customization capabilities play a significant role in determining competitiveness.
Who are the leading key players in the sterile injectable contract manufacturing market?
The leading key players in the sterile injectable contract manufacturing market are Baxter, Catalent, Inc., Vetter Pharma, Recipharm AB, Aenova Group, Fresenius Kabi, Unither Pharmaceuticals, Famar, Cipla Inc. and NextPharma Technologies.
Why do pharmaceutical companies use sterile injectable contract manufacturing?
Pharmaceutical companies use sterile injectable contract manufacturing to reduce costs, access specialized expertise, and focus on core competencies such as research and development.

